Lock-I
A simple device designed to standardize a procedure performed more than 20 million times a year worldwide.
In production
Status
23.04.2024
Entry date
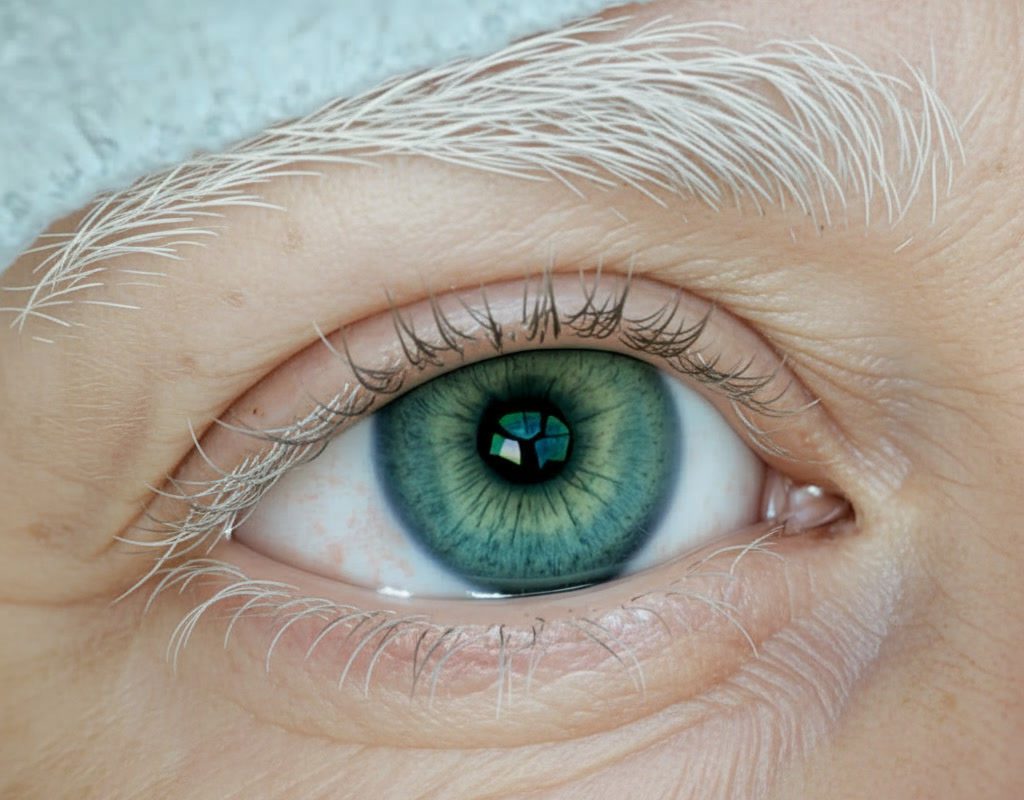
Problem to solve
A
Proposed solution
A
Request to H4
A

How this project is transforming healthcare : the problem to be solved
- Retinal diseases represent a global challenge, affecting more than 20 million patients who require intravitreal injections
- The positioning of the injection site currently depends on the operator
- This variability can lead to complications (e.g., lens touch) and a lack of reproducibility
- There is a need for simple standardization without complicating the procedure
The proposed solution
- Lock-I is a patented, sterile, single-use medical device
- It enables the injection site to be identified in a reproducible manner (3.5 mm / 4 mm depending on the condition of the lens)
- It does not alter the injection technique and integrates easily into current practice
- The goal is to improve safety, reproducibility, and training
Hands-On support from H4
- Project structuring and innovation positioning
- Support for strategic planning (regulatory, clinical, market)
- Connecting you with key experts and partners
- Preparation for project validation and next steps
What impact will this have on the rest of the project?
- Accelerating the project’s development
- Clarifying the next steps (clinical trial, academic partners)
- Strengthening the project’s credibility
